Plasma Medicine
Plasma medicine is an innovative field of research at the interface between physics and life sciences, which has experienced an immense international upswing in recent years. So-called cold atmospheric pressure plasmas are generated with the help of special plasma devices in order to achieve medical effects directly on or in the patient. The current main field of application is the treatment of chronic, infected wounds and pathogen-related skin diseases.
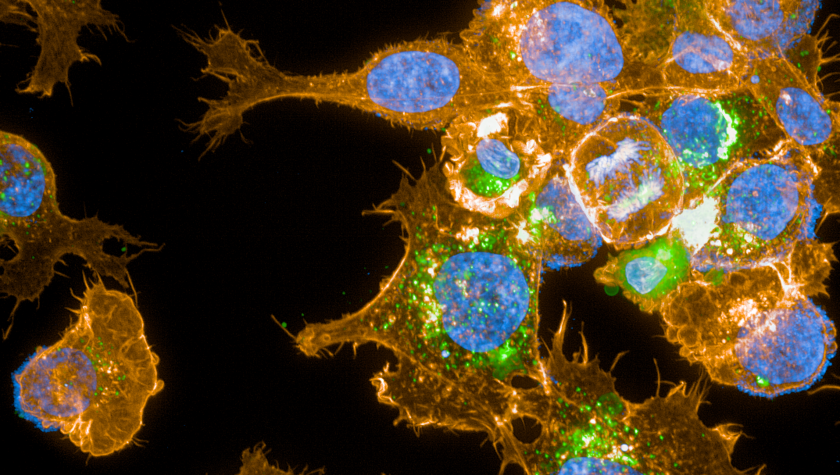
Plasma can be used to inactivate many types of microorganisms including multi-resistant bacteria such as MRSA. In addition, scientific and clinical studies show that plasma supports natural wound healing by stimulating tissue regeneration. Recent research results also indicate a potential benefit in the inactivation of tumor cells during longer or more intensive plasma treatments. This gives hope for new perspectives in cancer treatment. The underlying cell and molecular biological mechanisms of the interactions between plasma and living cells or tissue and their influence by modifying plasma parameters are being investigated at the INP by interdisciplinary teams. In particular, the investigation of plasma-generated reactive molecules, such as reactive oxygen- and nitrogen species (ROS), [TvW1] plays a central role. Another major goal of this work is the progressive characterization and control of the physical properties of cold atmospheric-pressure plasmas in interaction with living systems in order to redesign and optimize plasma devices for medical applications, thereby opening up new fields of application and putting plasma medicine on a solid scientific basis. In the course of this extensive research work, it has been demonstrated that the application of cold atmospheric-pressure plasmas is safe - an essential condition for their use in medicine.
The first cold plasma devices have already received market approval. The atmospheric-pressure plasma jet kINPen® MED developed at INP (CE-certified as a Class IIa medical device by neoplas med GmbH Greifswald in 2013) is successfully used for wound healing and in dermatology.
The research work is currently focused on the following topics:
- Investigation of redox-based mechanisms of biological plasma effects, identification of synergies between plasma medicine and redox biology
- Preclinical and translational research and support of clinical applications of cold atmospheric pressure plasma, especially in the fields of wound healing, cancer treatment and immunology
- Design, construction, experimental testing and optimization of atmospheric pressure plasma sources for special biomedical applications
The Research Program Plasma Medicine cooperates with various local, regional, national and international research partners and clinics. In the field of development and characterization of cold atmospheric-pressure plasma sources for medical applications, the Research Program is a partner for industry.
With its Research Program Plasma Medicine, INP is a member of various research associations:
- Leibniz Health Technologies (https://www.leibniz-healthtech.de/en/)
- Leibniz Research Network „Immune Mediated Deseases“ (https://www.leibniz-gemeinschaft.de/en/research/leibniz-research-networks/immune-mediated-diseases.html)
- NZPM - Nationales Zentrum für Plasmamedizin e.V. (http://www.plasma-medizin.de/)